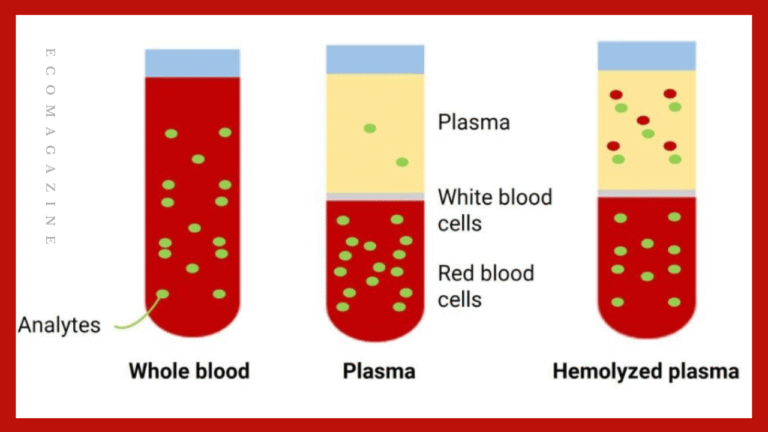
Hemolyzing in blood samples changes their composition and introduces errors that affect many laboratory tests. When red blood cells rupture, they release contents that interfere with measurements across clinical chemistry, hematology, and diagnostics. Understanding how hemolysis alters results helps labs prevent inaccurate reports, reduce repeat collections, and protect patient safety with consistent, high-quality analytical practices from start to finish.
How Hemolysis Alters the Composition of Blood Samples
Release of Intracellular Components Into Plasma or Serum
When red blood cells rupture, they release potassium, LDH, AST, and other intracellular components into plasma or serum. This sudden change alters the sample’s chemical balance and leads to false elevations. The release also affects protein distribution and enzyme activity. Understanding this shift helps labs identify hemolyzed samples early and prevents reporting values that no longer represent the patient’s true condition.
Chemical Interference From Hemoglobin and Cell Debris
Free hemoglobin and cell fragments introduce strong chemical interference. Hemoglobin absorbs light and disrupts photometric assays, while debris affects reaction kinetics. These components may bind reagents, distort color development, or cause incomplete reactions. This interference leads to unreliable results and complicates data interpretation. Recognizing how cell breakdown products influence assays helps teams decide when to reject or repeat testing.
Impact on Concentration Levels of Key Analytes
Hemolysis artificially elevates or reduces certain analytes. Potassium, phosphate, and enzymes rise due to intracellular release, while other markers may appear falsely low because of dilution or protein binding changes. These shifts distort clinical interpretation and may suggest conditions the patient does not have. Understanding these effects helps labs evaluate whether reported values reflect biology or sampling error.
Test Categories Most Affected by Hemolyzed Samples
Clinical Chemistry Assays Sensitive to Hemolysis
Clinical chemistry assays often rely on photometric detection, making them highly sensitive to hemoglobin interference. Tests for potassium, LDH, bilirubin, and AST show some of the largest shifts. Even slight hemolysis can alter absorbance curves or reaction speeds. Labs must monitor hemolysis indices and follow rejection criteria to avoid releasing inaccurate results that mislead clinicians and compromise patient management.
Hematology Measurements Influenced by Cell Destruction
Hematology tests depend on intact red cells, so hemolysis disrupts key measurements. RBC count and hematocrit drop artificially when cells rupture. Platelet and WBC counts may shift if debris interferes with detection systems. Morphology also suffers when samples become fragmented. Awareness of these effects helps technologists identify compromised samples and ensure hematology values reflect true patient status.
Diagnostic Markers With High Vulnerability to Artifacts
Some diagnostic markers, such as cardiac enzymes, troponin, and specific endocrine analytes, are highly vulnerable to artifacts caused by hemolysis. Free hemoglobin alters assay performance, while enzyme release masks true patient levels. These markers often guide critical decisions, so distorted results pose significant risk. Understanding their sensitivity helps labs apply strict review processes before reporting potentially misleading values.
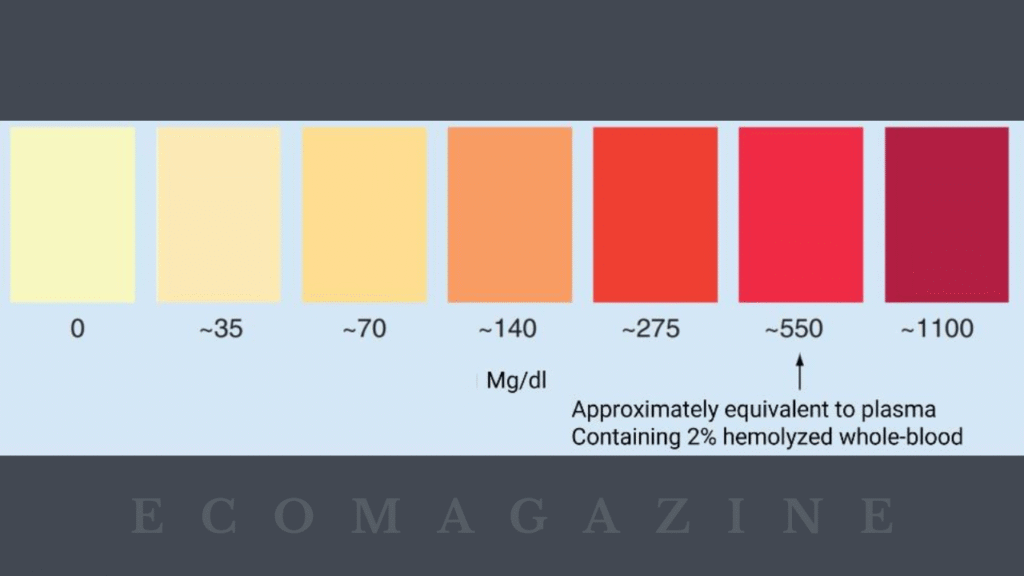
Mechanisms Behind Hemolysis-Driven Analytical Errors
Spectral Interference in Photometric Testing
Hemoglobin absorbs strongly at multiple wavelengths, causing spectral interference in photometric assays. This interference changes absorbance readings and distorts calibration curves. Even minor hemolysis may push results outside acceptable limits. By understanding this mechanism, labs adjust detection settings or flag impacted assays. Spectral interference remains one of the most common reasons hemolyzed samples produce unreliable chemical measurements.
Dilution Effects Caused by Cell Lysis
When cells rupture, the fluid portion of the sample expands slightly due to release of intracellular contents. This dilution alters concentrations of proteins and electrolytes. Some analytes appear lower than expected, while others mask changes due to overlapping effects. Recognizing dilution as a mechanism helps labs interpret unusual patterns and prevents misdiagnosis linked to inconsistent or unexpected values.
Enzyme and Electrolyte Distortion in Automated Analysis
Automated analyzers detect enzymes and electrolytes with high sensitivity. Hemolysis disrupts these measurements by releasing enzymes such as LDH and AST, and electrolytes like potassium. These distorted values may trigger false alarms for tissue damage or metabolic imbalance. Understanding how analyzers respond to hemolyzed samples helps labs validate results, prevent reporting errors, and ensure trustworthy clinical data.
Lab Practices That Minimize Hemolysis-Induced Bias
Clear Guidelines for Collection and Centrifugation
Standardized guidelines for venipuncture, tube handling, and centrifugation reduce hemolysis risk. Proper needle size, gentle mixing, and correct centrifuge settings protect cell integrity. Labs that enforce step-by-step protocols minimize variability and ensure consistent sample quality. Clear guidelines also help new staff avoid common mistakes that lead to hemolysis and maintain reliable workflows across all testing areas.
Quality Checks for Early Detection of Hemolyzed Samples
Visual inspection and automated hemolysis indices help labs detect problems before processing. Early detection prevents inaccurate reporting and reduces unnecessary repeat testing. Quality checks also highlight patterns that reveal training or equipment issues. By reviewing hemolysis rates regularly, labs track performance, strengthen procedures, and keep sample integrity at the center of every testing decision.
Consistent Workflow Controls to Reduce Variability
Strong workflow controls reduce the variability that leads to hemolysis. Standardized transport, temperature management, and processing timelines protect samples from stress. Regular equipment maintenance ensures instruments handle samples gently. When labs maintain consistent, controlled workflows, they limit opportunities for hemolysis to occur and create a dependable environment that supports accurate, reproducible testing across departments.
Conclusion
Hemolyzing in blood samples introduces chemical interference, distorts analyte levels, and disrupts accuracy across many test categories. When labs understand how blood hemolyzing drives analytical errors, they can strengthen protocols and reduce bias. Clear guidelines, early detection tools, and controlled workflows protect sample integrity. A proactive approach ensures clinicians receive dependable results and patients benefit from informed, reliable medical decisions.